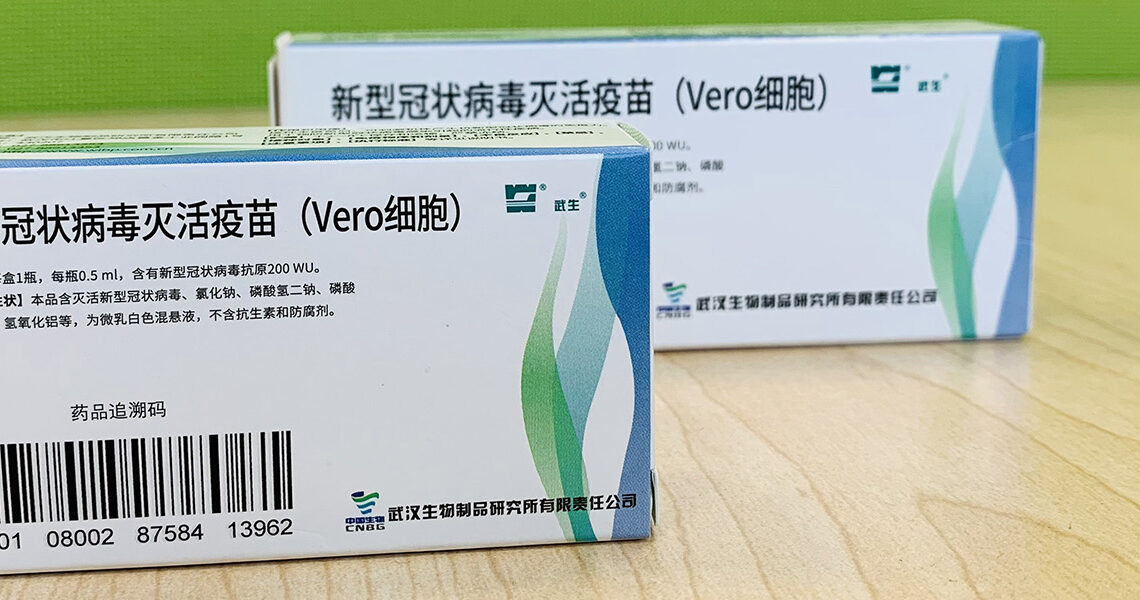
Metsä Board’s paperboard protects COVID-19 vaccines
April 28, 2021Good hygiene and cleanliness, as well as strength
and sturdy attributes make Metsä Board’s paperboards suitable for all kinds of
demanding healthcare packaging such as vaccines. MetsäBoard Pro FBB Bright 245
g/m2 has been approved by the Chinese pharma companies Sinovac (Beijing) and
Sinopharm (Wuhan) for use in their COVID-19 vaccine packaging. The lightweight,
yet robust, vaccine packaging comes in two size options – one for the COVID-19
vaccine bottle only, and one for the COVID-19 vaccine bottle and the
injector.
“MetsäBoard Pro FBB Bright is of a consistently high quality and its
outstanding performance has resulted in many highly satisfied customers. The
most impressive part is its smooth runnability for printing, die-cutting and
gluing processes and even on automatic packing lines at a speed of 500 boxes
per minute,” said responsible person of production from the converting
company.
“It’s an honour for Metsä Board to be actively contributing its
share to combating the pandemic”, commented Andy Zhong, Metsä
Board’s local sales manager. “It is a strong board which is vital to
protect the delicate contents and made from lightweight, pure fresh fibre
making it ideal for pharma packages that require purity.”
MetsäBoard Pro FBB Bright is a premium lightweight folding boxboard made with
100% pure fresh fibre from renewable, traceable and sustainably managed
northern forests, and it is available with PEFC™ or FSC® certification. The Product
Safety Statement indicates that the paperboard is in compliance with relevant
global product safety related regulations and recommendations.

